
The tables, from the second one, summarize the recommended data expected to be useful for plasma modelling in fusion. Journal of Applied Mathematics and Physics,ĪBSTRACT: This paper presents a review about the radiative properties (transition probabilities and oscillator strengths) of two xenon ions (Xe9+, Xe10+) and three members of Er I isoelectronic sequence (Lu3+, Hf4+, Ta5+) of interest in controlled thermonuclear fusion, including our recent theoretical data obtained using two independent theoretical atomic structure computational approaches (semi-empirical Hartree-Fock with relativistic corrections method (HFR) and the ab initio multiconfiguration Dirac-Hartree-Fock (MCDHF)). Lines shown in grey are not detectable by the XGT instruments. The aim of this work is to provide Stark parameters of low intensity Xe II and Xe III spectral lines from which scarce or null previous information exist.This work completes the measurements of ionized Xenon parameters developed in the last few years and extends Stark parameters database for astrophysical applications. Recent Progress in Radiative-Rate Determination of Some Heavy Ions (Xe9+, Xe10+, Lu3+, Hf4+, Ta5+) of Interest in FusionĪtomic Spectra, Atomic Data, Transition Probabilities, Oscillator Strengths, Heavy Elements This applet was successfully run under the following browsers : NetScape Navigator 3.01. (b) The energy of the orbit becomes increasingly less negative with increasing n. 2: The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Journal of Physical and Chemical Reference Data, 33, 765-921. The simulated gas discharge spectrum is synthesized by assigning each emission line to a gaussian and each point in the spectra is computed as a mathematical sum of all the emission lines. In this state the radius of the orbit is also infinite. Download scientific diagram The original spectrum lines of Xenon (a) and Helium (d) and the normalized lines were decomposed to the primary colors (b. (1983) Energy Levels and Observed Spectral Lines of Xenon, Xe I through Xe LIV. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.Saloman, E.B. The energy levels and observed spectral lines of the xenon atom, in all stages of ionization for which experimental data are available, have been compiled. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii.  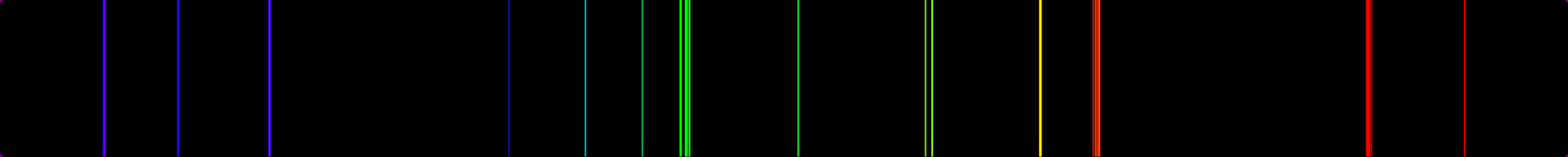
In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Lamp Emission Spectra Xenon Compact Arc Lamps Wavelength, nm output Wavelength, nm output Wavelength, nm output Wavelength, nm output 250-300 2.489 750-800 4.682 1250-1300 0.896 1750-1800 0.345 300-350 3.540 800-850 8.914 1300-1350 0.705 1800-1850 0.306 350-400 4.577 850-900 6.284 1350-1400 0.931 1850-1900 0. Formula: Xe Molecular weight: 131.293 IUPAC Standard InChI:InChI1S/Xe IUPAC Standard InChIKey:FHNFHKCVQCLJFQ-UHFFFAOYSA-N CAS Registry Number: Chemical structure: This structure is also available as a 2d Mol file Other names: Xe UN 2036 UN 2591 Xenon atom Xeneisol 133A Xenomatic Permanent link for this species.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
